Want to chat with us during AACC?
We want to see you in Chicago. Use the link below to schedule time with EDP during the show.
We’d love to schedule some time to meet you at AACC next week. In attendance will be Eric Mayer, CEO, Jon Dancy our Business Development Manager, and Mark Boyd, our Marketing Manager.
EDP is a full-service IVD company, and we are exploring new partnerships at AACC to grow our CLIA lab test menu, product development pipeline, and analytical+clinical study services.

What does EDP do?
Full Service IVD/CDx CRO – Strategy, Product Development, Regulatory Approvals, and Commercialization across multiple platforms and therapeutic areas:
- Strategy, Product Development, Regulatory Approvals, and Commercialization
- multiple platforms and therapeutic areas
- Get your diagnostic/device to market faster
We know the FDA, and what it takes to get to Market.
- Over 75 products submitted and approved by the FDA
- We keep up-to-date on the protocol changes, so your device or design doesn’t run into unforeseen issues due to technicalities
- We simply love this stuff.

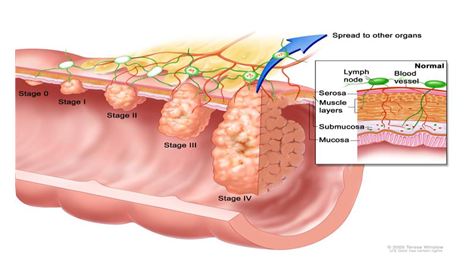
- Most recent research study results for CRC and polyp detection
- Upcoming publication on biomarker utility
We’re looking forward to seeing you at the Show!